Dr_Microbe/iStock by way of Getty Photographs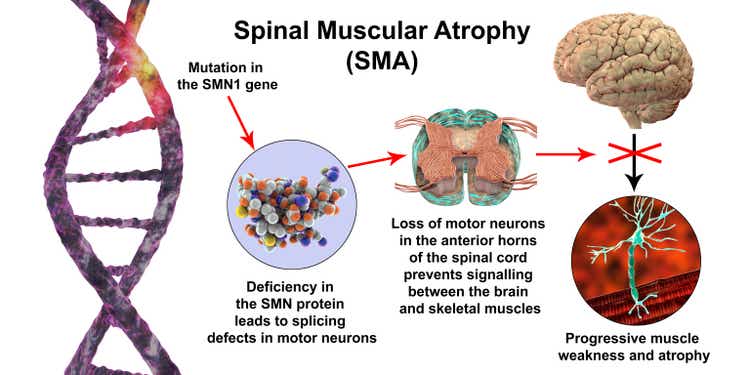
Roche’s (OTCQX:RHHBY) (OTCQX:RHHBF) Genentech claimed new long time period info for for Evrysdi (risdiplam) in spinal muscular atrophy (SMA), an inherited disorder characterized by reduction of nerve cells top to muscle weak spot over time.
The enterprise reported a few-12 months info from the SUNFISH research verified increases in motor purpose are sustained at a few many years whilst adverse situations lessened above the similar interval in people today aged 2-25 several years with Variety 2 or Sort 3 SMA.
The business said Evrysdi was effectively-tolerated around the 3 a long time. The all round level of adverse situations (AEs) decreased about a few decades, and a development towards a reduced amount of severe adverse events (SAEs) was noticed in the 3rd year of cure.
AEs and SAEs ended up reflective of the underlying sickness and no treatment-connected AEs led to withdrawal from the review.
In addition, the company described exploratory two-calendar year efficacy information from Section 2 of SUNFISH review, displaying improvement in, or stabilization of motor purpose with Evrysdi in comparison to an untreated external manage team.
Genentech also described newest interim success from the RAINBOWFISH review which showed that the the greater part of babies addressed with Evrysdi for at minimum 12 months were in a position to stand and walk inside timeframes regular of healthier infants.
In January, Fda granted precedence evaluate to Evrysdi for pre-symptomatic newborn toddlers with SMA.
